The key lies in indicators: Interlab redefines management in the Sterile Processing Department
Digital transformation in healthcare does not begin or end with the electronic medical record. It also reaches critical and often invisible areas that uphold patient safety at its foundation. One of them is the Sterile Processing Department. There, where every processed tray directly impacts a surgical procedure, indicator-based management is no longer an aspiration — it has become a strategic necessity.
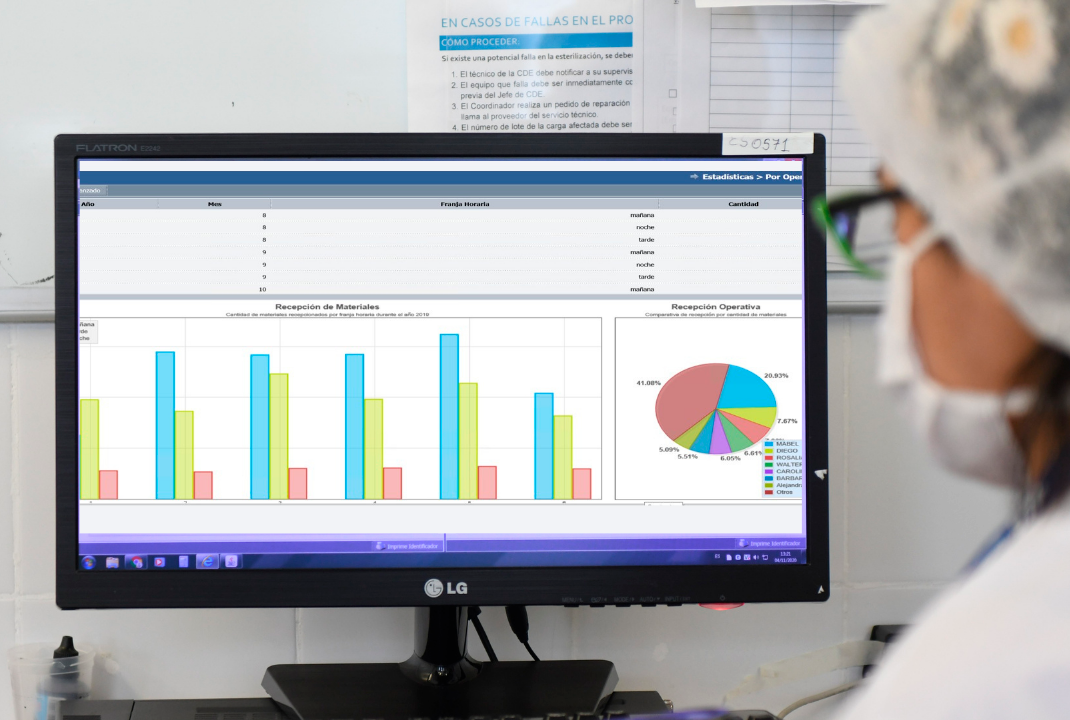
In this context, Interlab’s traceability and management software introduces a paradigm shift: it turns daily operations into structured, measurable, and actionable information. In doing so, it redefines the role of the Sterile Processing Department within the institution.
For years, the department’s efficiency was measured by its ability to meet surgical demand and comply with protocols. However, in an increasingly demanding healthcare system — shaped by audits, accreditations, and quality standards — compliance alone is no longer enough. Today, institutions must demonstrate, anticipate, optimize, and support decisions with evidence.
Each stage of the process generates critical data: receipt of contaminated instruments, washing, tray assembly, chemical and biological controls, cycle validation, sterile release, and distribution. It is a complex circuit where the margin for error is minimal and full traceability is essential. Yet when this information remains scattered across manual records or non-integrated systems, its strategic value is diluted.
The differentiating factor of Interlab’s solution lies in integrating all traceability into a single platform and automatically converting it into clear, comparable, and auditable indicators. Response time, reprocessing rates, assembly deviations, control compliance, and shift productivity cease to be perceptions and become objective metrics.
This comprehensive visibility changes the management logic. The Sterile Processing Department no longer acts only on what has already occurred — it can anticipate. When an indicator shows variation, the system precisely identifies where in the process the deviation originated. Management becomes preventive. Decisions rely on consolidated data. Risks are reduced before they escalate.
In contexts where institutions pursue international accreditations or must respond to internal and external audits, having verifiable information is decisive. The software not only documents every cycle and validation but also supports each action with full traceability and accessible reporting. Patient safety is thus sustained by systematized and reliable information.
By integrating KPIs within the same tool that manages operations, Interlab transforms daily work into measurable performance. Hospital leadership can analyze trends, detect improvement opportunities, plan resources more accurately, and make evidence-based decisions. The Sterile Processing Department ceases to be an invisible area and becomes a strategic actor capable of demonstrating tangible results.
In this scenario, innovation is not merely about digitalizing processes. It is about transforming data into decisions and decisions into continuous improvement. That is the leap Interlab proposes: elevating sterilization management to a new level, strengthening safety, and supporting healthcare institutions in their evolution toward increasingly intelligent, information-driven models.



